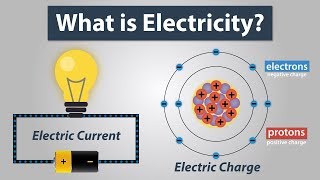
Whenever we rub two objects together, they become electrically charged. This is because atoms divisible as they consists of charged particles.
Therefore, atoms can be further divided into proton, neutron and electron.
By 1900, it was known that the atom is not a simple and indivisible particle but contains at least one sub-atomic particle – the “electron”. This theory was proposed by J.J. Thomson.
He carried a “cathode Ray” experiment in which observed a stream of negatively charged particle coming out of cathode towards the anode, which ultimately known as electron.
In 1886, E. Goldstein discovered the presence of new radiation in a gas discharge and call them “canal rays” which were positively charged radiation.
This was observed in the same gas discharge tube, emitting positive particles from anode.
This ultimately led to the discovery of another sub-atomic particle which had a charge equal in magnitude but opposite in sign to that of electron and mass approximately 2000 times as that of the electron. It was given the name “proton”.
An electron is represented as ‘e–’on a proton as ‘p+’.
Neutrons were discovered by charge “James Chadwick” in 1932.
Atoms are made up of protons and electrons in equal or balanced proportion.